Last updated 9/2020
MP4 | Video: h264, 1280x720 | Audio: AAC, 44.1 KHz
Language: English | Size: 1.61 GB | Duration: 6h 31m
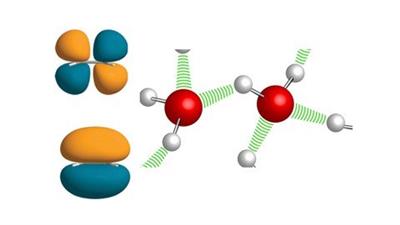
Wave Mechanical Concept of the Covalent Bond
What you'll learn
Wave Mechanical Concept of the Covalent Bond - Valence Bond Theory, Molecular Orbital Theory and VSEPR Theory.
Techniques to construct Molecular Orbital diagrams.
Bond Order calculation for a variety of Homonuclear and Heteronuclear molecules.
Predicting molecular geometries using VSEPR Theory.
Requirements
Basic Knowledge of Chemistry.
Description
This course covers the theories related to the Wave Mechanical Concept of the covalent bond beyond Lewis theory.The following are the highlights of the course -A brief idea about wavefunction.Valence Bond Theory - Its postulates and limitations.Molecular Orbital Theory -What is M. O. Theory?Conditions for the combination of Atomic Orbitals - The LCAO concept.Effective overlap of Atomic Orbitals - The symmetry conditions.Types of Molecular Orbitals and how we obtain them? (sigma and pi M. O.)Distribution of Molecular Orbitals and its discrepancy - Orbital mixing.The concept of s-p Mixing.Bonding and Antibonding M. O.s - What do they mean?Construction of Molecular Orbital Diagrams - How to draw M. O. diagrams?Homonuclear molecules - H, He, Li, Be, B, C, N, O, F and Ne molecules;Various types of Oxygen ions.Heteronuclear molecules - The concept behind heteronuclear M. O. diagram construction.CO, NO and HF.Calculation of Bond Order.Comparative analysis of Valence Bond and Molecular Orbital Theories.The VSEPR concept - Salient FeaturesRelation with Hybridization, Bond angle, and Electron Pairs.Applying VSEPR theory to -Molecules with regular geometry -BeF2, BCl3, CH4, PF5, SF6, IF7Molecules with irregular geometry -IF5, ClF3, SF4, XeF2, I3-, XeOF2Tips and tricks to flawlessly predict molecular geometries using VSEPR.Applying VSEPR concept - explained with few practice problems.
Who this course is for
Students of 11th and 12th grade in Science stream.,Students pursuing Bachelor's degree with Chemistry as one of the subjects.,Students pursuing Master's in Chemistry.,Students preparing for various competitive exams.
Download link
rapidgator.net:
uploadgig.com:Code:https://rapidgator.net/file/bff16be43d87894c18f99d5760d7c1b8/yiozu.Advanced.Structure.And.Bonding.In.Chemistry.part1.rar.html https://rapidgator.net/file/eb8a56309f17ce7aee2097bdf0ed6e41/yiozu.Advanced.Structure.And.Bonding.In.Chemistry.part2.rar.html
nitroflare.com:Code:https://uploadgig.com/file/download/066652f195BcDe22/yiozu.Advanced.Structure.And.Bonding.In.Chemistry.part1.rar https://uploadgig.com/file/download/Ac1b84C520A56557/yiozu.Advanced.Structure.And.Bonding.In.Chemistry.part2.rar
1dl.net:Code:https://nitroflare.com/view/E3C286222D6216F/yiozu.Advanced.Structure.And.Bonding.In.Chemistry.part1.rar https://nitroflare.com/view/EEFF55328A7F500/yiozu.Advanced.Structure.And.Bonding.In.Chemistry.part2.rar
Code:https://1dl.net/ddicd9om611g/yiozu.Advanced.Structure.And.Bonding.In.Chemistry.part1.rar.html https://1dl.net/wk3t5xbg32o6/yiozu.Advanced.Structure.And.Bonding.In.Chemistry.part2.rar.html




 Reply With Quote
Reply With Quote